|
|
[交流]
单靠实验数据统计拟合能逼近真理吗? ——论“凭什么相信计算”之三 已有5人参与

无机物化版块专家(mbchen)活动
![单靠实验数据统计拟合能逼近真理吗? ——论“凭什么相信计算”之三]()
原载:化学通报, 2016, 79: 196-204.
单靠实验数据统计拟合能逼近真理吗?
——论“凭什么相信计算”之三
陈敏伯
(中国科学院上海有机化学研究所 上海 200032)
摘要 鉴于诸如物理有机化学领域改进Hammett方程等引起的学术争论,本文以气体状态方程、电负性和硬软酸碱原理为例,分析在科学求真道路上得到的教训和成功经验,说明单靠增加实验数据统计拟合来“改进”唯象理论、以期逼近真理的做法是没有根据的。历史上也没有成功的先例。这种做法在统计意义上是正确的,但与物理意义上的正确决不是一回事。
关键词 唯象理论,实验数据拟合,气体状态方程,电负性,硬软酸碱理论
=======================================
(正文)
“凭什么相信计算”的问题,实际上是在问凭什么相信计算所依据的理论。本文要讨论的是单靠实验数据统计拟合能否不断逼近真理。这里的“逼近真理”不是哲学意义上的,而是科学意义上的,即:一个好的科学理论无非是对解释或理解自然达到最大范围统一和自洽的说理体系[1]。
根据实验结果拟合得到的规律称为唯象理论(phenomenological theories)。单靠实验数据拟合就是单靠唯象理论。唯象理论的例外总是容易找到的。其中,唯独热力学几乎找不到例外。其原因除了人们长期寻找第一类和第二类永动机的实验企图都遭到失败之外,还因为人们发现热力学理论在一定层次上具有公理结构[2]。
§1 从改进Hammett方程的争论谈起
实际上,长期以来持有单靠实验数据拟合就能逼近真理的观点在化学界大有人在。例如,Hammett方程是关于取代基对一系列化合物的反应速度常数或反应平衡常数的影响程度的唯象方程。1937年Hammett靠实验数据拟合提出了这个方程[3],它的提出促成了物理有机化学这门三级学科的建立。尽管至今已经70多年,还有很多化学家致力于不断改进它[4]。寄希望通过更多次地改进Hammett方程来逼近真理。但是,至少从上世纪60年代开始就有强烈、公开的否定意见出声[5]。
持赞成意见的人们说(2003年):“尽管Hammett方法由于其经验性而受到理论化学家的批评,但令人吃惊的是,简单地从溶液中有机酸的电离获得的σ常数,常常能够成功地预测多种系列反应的平衡和反应速率常数” [4]。他们认为:增加实验数据,加上定性或半定量的直观化学理解,添加Hammett方程中的参数数目,运用统计数学的线性或非线性回归方法来调节参数数值,只要相关系数又很高,满足置信度要求,那就可以改进Hammett方程。不断改进下去就能逼近真理。有什么不妥呢?。
在反对意见那一边,如1969年M J S Dewar教授尖锐地批评道[5]:“现在可供利用的这种数据近乎有1000种系统。其奥秘归结到环绕方程(9.102)【注:指log k = ρσ,其中k为反应速度常数或平衡常数;ρ和σ为引入的参数】,不过当体系不服从方程(9.102)时,许多化学工作者觉得必须用相宜的修改来纠正这些缺陷,各种σ常数集合( , , , , , , 等)的完整系列发展到使顽固派纳入到正轨,结果他们的处境是十分尴尬的。回顾起来,难以理解为什么这样多的努力要投进这样一个完全没有收获的领域里”。Dewar这里所指的“相宜的修改”就是指把Hammett方程从两个待定参数的log k = ρσ “改进”为例如 等)的完整系列发展到使顽固派纳入到正轨,结果他们的处境是十分尴尬的。回顾起来,难以理解为什么这样多的努力要投进这样一个完全没有收获的领域里”。Dewar这里所指的“相宜的修改”就是指把Hammett方程从两个待定参数的log k = ρσ “改进”为例如 之类含更多待定参数的方程,或其他函数形式的方程。笔者没有查证当时此类改进方案是否如Dewar所说已近乎千种;不过,哪怕有五十种,难道还不足以让人反省吗。Dewar反对的正是那种企图单凭实验数据拟合来逼近科学真理的做法。 之类含更多待定参数的方程,或其他函数形式的方程。笔者没有查证当时此类改进方案是否如Dewar所说已近乎千种;不过,哪怕有五十种,难道还不足以让人反省吗。Dewar反对的正是那种企图单凭实验数据拟合来逼近科学真理的做法。
有人会辩护说:Hammett方程在热力学上有线性自由能关系作其理论基础。不错,但仔细分析就可以看出这样的辩护是没道理的。理由是,任意体系只要不是远离平衡的、爆炸性的,即数学上不是发散的,哪怕收敛很慢,该体系的任意一种性质总是可以在平衡态附近展开为Taylor级数,线性关系不过就是保留一次项捨去二次及以上项而已,无论是自由能,还是什么性质,都是成立的,不足为奇。可是,这只限于线性关系,线性自由能关系显然不能成为通过纳入高阶项来逼近真值的根据。
还有人会辩护说:改进Hammett方程所选用的数据都是有选择的,选择的依据是不同取代基在各种化学反应中的差别,这种差别或是定性的,或是半定量的,是有物理意义的。其实这样的辩护也是乏力的。他们所说的物理意义将与以下2、3、4节所述的类似,只能说是若隐若现的,没有靠得住的严格(而不只是定量)的概念作后盾。
化学领域里建立和“改进”此类经验规律即唯象理论的例子为数极大,远远不止Hammett方程一例。在物理有机化学、化学化工、甚至其他工程领域中都可以看到无数个如此的方程,占据了目前化学“理论”的绝大多数[6]。
随着上世纪90年代计算机技术提供的强大助推力,单靠实验数据拟合探求真理的做法又被推上高潮。试图通过人工智能、海量实验数据的高速处理来探求化学真理。例如用化学信息学、化学计量学、数据挖掘方法(data mining)探求化学真理,例如红外光谱的谱线位置、核磁共振的化学位移以及探求各种结构-性质关系(即所谓“构效关系”)的统计模型等等。但本质上,还都是企图单凭实验数据拟合来逼近科学真理的做法。
尽管两千多万种化合物在物质本原上是统一的,即化合物都是由不多几种原子核和数目不等的电子组成的,但是这些唯象理论却各据一方,各说各的道理,互相很少联系。即使这样的“理论”有时对工程问题有用,但是它们之间既不统一,也不自洽。“有用”与逼近真理远不是一回事。
任何学科在幼年时期总是从同样的做法开始:将自然界中的所有对象分门别类、然后作对比。此后,有的学科进一步采用数理统计方法把科学认知半定量化、甚至定量化,得到唯象理论。例如Pauling电负性公式[7]、反应速度的Arrhenius定律[8]、反应速度常数或平衡常数中的Hammett方程[3]、电学中的Ohm定律、扩散中的Fick定律、导热中的Fourier定律、流体粘度的Newton定律、实际气体的van der Waals公式[8]……都是唯象理论。唯象理论是真正科学的起点,是通向真理的中间站。尤其可贵的是那最早的一、两种唯象理论。现在的问题是:不能夸大唯象理论的意义,如果不依靠演绎法,即使补充新的实验数据、将唯象理论改进一千次也无法逼近真理。历史上没有一个成功的先例。
为此,回顾更多个重要化学概念不断深化的历史过程是很有启发作用的。从中可以悟出一些道理:看看唯象理论距离真理还有多远;改进唯象理论的出路何在;是否单靠实验数据拟合,就能逼近真理?具体分析以下三个理论或概念:气体的状态方程、元素的电负性,和硬软酸碱原理(HSAB原理)。它们的演化历史与Hammett方程等其他唯象理论走过的道路十分相像。都曾经从最初几个唯象理论开始,经过了漫长的单靠实验数据拟合的“改进”;但不同之处在于有的已经或接近走出了迷宫,最后确立了基于第一原理的普适概念;但是其余为数更多的唯象理论目前还没有走出迷宫,有的还不知道身在迷宫中。
§2 实际气体状态方程的演化
历史上,气体理论的研究与最简单的力学问题一样是整个现代化学、物理学的发端。1834年Clapeyron将Boyle定律(1662年)和Charles - Gay-Lussac定律(1787年)结合起来得到理想气体的状态方程
 ,或 ,或 , (1) , (1)
其中p、V、T、n和R分别为气体的压强、体积、温度、摩尔数和气体常数; 为气体的摩尔体积。理想气体状态方程是为气体提出的第一个唯象理论。只要在温度不低和稀薄的条件下任意气体都相当准确地遵守式(1)。1873年,van der Waals将理想气体方程改造成在其他条件下任意气体也适用的状态方程,即所谓实际气体的状态方程 为气体的摩尔体积。理想气体状态方程是为气体提出的第一个唯象理论。只要在温度不低和稀薄的条件下任意气体都相当准确地遵守式(1)。1873年,van der Waals将理想气体方程改造成在其他条件下任意气体也适用的状态方程,即所谓实际气体的状态方程
\left ( v-b \right )=RT) , (2) , (2)
只是其中引入两个参数a、b,它们取决于气体的具体种类。van der Waals气体方程在理论上最大进步是对这两个参数a、b似乎说得出一点物理意义: 相当于当分子间距太近时分子间的排斥力造成分子所感受到的实际压强要比测 量的压强p来得大一点;b相当于摩尔气体的“硬芯”体积,即气体分子感受到的体系体积应当要比外部测量的体系体积小些。 相当于当分子间距太近时分子间的排斥力造成分子所感受到的实际压强要比测 量的压强p来得大一点;b相当于摩尔气体的“硬芯”体积,即气体分子感受到的体系体积应当要比外部测量的体系体积小些。
姑且先不追究van der Waals气体这样说法的物理意义其严格程度到底如何。无论如何,理想气体方程和van der Waals气体方程是关于任意气体的最早、最简单而有一定物理意义的唯象理论。它们非但奠定了现代气体理论的基础,实际上也是为现代自然科学基础奠定了重要的一部分。此后提出的的改良方案基本上都是依此为出发点的,见表1。
还有许多其他的气体唯象理论没有包括在表1内,如Redlich–Kwong-Soave方程(1972年)、 Peng–Robinson–Stryjek–Vera方程(PRSV,1986年)、PRSV2方程(1986年)、Elliott-Suresh- Donohue方程(1990年)等[9]。
表1 气体状态方程的几种唯象理论[8,9,10]
名称 状态方程 年代
理想气体  1834年 1834年
van der Waals气体 \left ( v-b \right )=RT) 1873年 1873年
Clausius气体 ^{2}} \right \}\left ( v-b \right )=RT) 1880年 1880年
Berthelot气体 \left ( v-b \right )=RT)
Dieterici气体(A) \left ( v-b \right )=RT)
Dieterici气体(B) =RT)
某实际气体模型 \left ( v-b \right )=RT)
Redlich–Kwong气体 v} \right )\left ( v-b \right )=RT)
1949年
Peng-Robinson气体 \left ( v-b \right )=RT)
其中: \right )^{2}) ;对比温度 ;对比温度  , ,
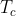 临界温度; 临界温度; , ,
因子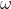 取决于具体物种。 1976年 取决于具体物种。 1976年
virial展开(Mayer夫妇) }{v}+\frac{C\left ( T \right )}{v^{2}}+... \right \}) 1940年 1940年
细细对比这些方程,读者你也能悟出怎么“改进”的奥妙来,正如Hammett方程改进时一样,其中也都能说出若隐若现、不甚确凿的物理意义来。如Elliott-Suresh-Donohue方程申称是修正van der Waals方程的排斥项后得到的。但是,沿着这条思路下去,读者也会对这么多的“改进”发生怀疑。因为所谓改进无非是增加实验数据,增加方程中的参数数目,调节参数,使线性或非线性拟合的统计指标(如相关系数R,或方差比) 等)达到人为指定的置信度,如90%或95%。对于增加的几类反应数据的拟合的确改进了,但同时对没有收集到实验数据的几类反应的结果往往反倒变坏;单靠统计不靠物理,这种捉襟见肘的现象是无法避免的。越改进,参数的物理意义也越模糊。除了virial气体理论之外,所有的唯象理论都无法覆盖无穷多种实际气体的。所以,除了最早的一两个唯象理论之外,其余的“改进”实在是没多大意义的。除非是例如工厂生产某气体产品的需要,才会再去搞一个新的状态方程,否则都会陷入统计数学模型的泥潭里,越陷越深。 等)达到人为指定的置信度,如90%或95%。对于增加的几类反应数据的拟合的确改进了,但同时对没有收集到实验数据的几类反应的结果往往反倒变坏;单靠统计不靠物理,这种捉襟见肘的现象是无法避免的。越改进,参数的物理意义也越模糊。除了virial气体理论之外,所有的唯象理论都无法覆盖无穷多种实际气体的。所以,除了最早的一两个唯象理论之外,其余的“改进”实在是没多大意义的。除非是例如工厂生产某气体产品的需要,才会再去搞一个新的状态方程,否则都会陷入统计数学模型的泥潭里,越陷越深。
Mayer夫妇的virial气体理论有其独特的学术价值[10]。尽管按密度展开即virial展开粗看是数学技巧,但是通过统计力学的巨配分函数原则上可以以任意精度导出任意实际气体的各阶virial系数,B_{2}\left ( T \right ),...) ;更重要的是可以直接与气体分子间的微观相互作用联系起来(见[11]的第5.5节)。这就在理论上体现了质的不同,任何唯象理论无论怎么改进都做不到这一点。这样Mayer理论就超越了唯象理论,进入严格理论的范畴,尽管还不够臻美。 ;更重要的是可以直接与气体分子间的微观相互作用联系起来(见[11]的第5.5节)。这就在理论上体现了质的不同,任何唯象理论无论怎么改进都做不到这一点。这样Mayer理论就超越了唯象理论,进入严格理论的范畴,尽管还不够臻美。
从理想气体方程和van der Waals气体方程这两个最早的唯象理论直到1940年Mayer的严格理论之间的七十年间,曾经出现过多达150种不同的唯象改进方案[12]。很难说清这150多种方程有多少实质性的进步。这点与Hammett方程改进的历史如出一辙。它们只是不同的数学模型而已,想用统计回归中的不同种统计指标来表征有“改进”,实在缺乏说服力。只有到了Mayer理论才有了实质上的进步。这种进步是物理上的,不是统计上的。
最后的命运是:今天人们只须记住三个气体理论:理想气体方程、van der Waals方程,和Mayer的集团展开理论。其余的状态方程忘了也无大碍。与此相比,可预见Hammett方程改进的命运也不会有质的不同。
§3 电负性概念的演化
第二个实例是电负性 ,这是化学中少数几个最基本的概念之一。目前大多数化学家头脑中的电负性概念是L. Pauling在1932年提出的[7]。人们把它“理解”为:表征分子中原子对电子的吸引能力。此后还有许多关于“改进”电负性概念的方案[13-20]。直到1978年,Parr提出绝对电负性的概念,真正与热力学、统计力学、量子力学融为一体,其物理意义既清晰又严格,终于一锤定音[21]。这是第一个严格地在第一原理基础上诞生的化学概念。 ,这是化学中少数几个最基本的概念之一。目前大多数化学家头脑中的电负性概念是L. Pauling在1932年提出的[7]。人们把它“理解”为:表征分子中原子对电子的吸引能力。此后还有许多关于“改进”电负性概念的方案[13-20]。直到1978年,Parr提出绝对电负性的概念,真正与热力学、统计力学、量子力学融为一体,其物理意义既清晰又严格,终于一锤定音[21]。这是第一个严格地在第一原理基础上诞生的化学概念。
Pauling对元素电负性描述来自同核双原子分子和异核双原子分子的键能比较:设想将双原子异核分子B-C(B、C代表任意原子)拆散成B原子和C原子,需要外界加入的能量为键能D(B-C)。倘若把D(B-C)看成同核双原子分子键能D(B-B)和D(C-C)的平均值显然相当粗糙,需要修正。即
D(B-C) = [ D(B-B) + D(C-C) ] / 2 + (修正项)。
修正项要满足在同核双原子分子时等于零。可以想象修正项与分子中原子B和C对电子吸引能力的差别有关。Pauling把这种分子中原子对电子的吸引能力称为“电负性” 。令 。令 和 和 分别表示B、C原子的电负性。假设在所有各种分子中同一原子的 分别表示B、C原子的电负性。假设在所有各种分子中同一原子的 都近似相同。形式上还须满足D(B-C) = D(C-B),于是总可写成 都近似相同。形式上还须满足D(B-C) = D(C-B),于是总可写成
D(B-C)=0.5*[D(B-B)+ D(C-C) ] + k(χB – χC)^2 ,
其中k为待定常数。以3种元素(如F, Cl, Br)为例,有上述的方程3个,要解4个未知数 、 、 、 、 和k显然不可能,故需人为约定一个元素的电负性,如氟, 和k显然不可能,故需人为约定一个元素的电负性,如氟, ,继而得到 ,继而得到
D(B-C)=0.5*[D(B-B)+ D(C-C) ] + 23(χB – χC)^2
(单位 ) (3) ) (3)
这就是Pauling的电负性定义[7]。虽然,Pauling电负性实际上不是分子中原子对电子吸引能力的绝对量度,而是相对量度;但Pauling电负性是这方面第一个唯象理论,极为重要,无论有多少缺点,也无损它的历史地位。
表2 历史上出现过的电负性定义的典型方案[7,13-21]
名称 电负性定义 年代,文献
Pauling的电负性 D(B-C)=0.5*[D(B-B)+ D(C-C) ] + 23(χB – χC)^2
设 1932, [7] 1932, [7]
Mulliken的电负性 ) 1934, [14] 1934, [14]
Gordy的电负性  1946, [15] 1946, [15]
李世瑨的电负性  1957, [16] 1957, [16]
Allred-Rochow电负性  1958, [17] 1958, [17]
高孝恢的电负性 
1961, [18]
温元凯的电负性  1974, [19] 1974, [19]
Parr的绝对电负性 _{v})
(E,N为体系的能量和电子总数) 1978, [21]
Allen的电负性  1989, [20] 1989, [20]
*:N为原子的价电子数, 为有效核电荷,r为原子的共价半径, 为有效核电荷,r为原子的共价半径, 为离子半径,e为质子或电子的电荷量, 为离子半径,e为质子或电子的电荷量, 为第i号价电子的有效主量子数, 为第i号价电子的有效主量子数, 为游离原子中s,p电子的能量, 为游离原子中s,p电子的能量, 为游离原子价电子层中s,p电子的个数。v外势,从电子来看外界对它造成的势能。其余变量说明见正文。 为游离原子价电子层中s,p电子的个数。v外势,从电子来看外界对它造成的势能。其余变量说明见正文。
在此后的半个世纪里,为了改进Pauling的电负性曾经出过几十篇“改进方案”的论文[15-20]。他们走的道路与改进Hammett方程、改进气体状态方程走过的道路一样,都是靠实验数据累积、加入更多参数、采用不同的数学模型,最后统计拟合(见表2)。结果可想而知,都跳不出“Russell鸡”的逻辑[22]。所以它们的历史地位远不如Pauling。
与众不同的只有两位:一是1934年Mulliken根据光谱数据提出的电负性定义[14]
) , (4) , (4)
其中I和A分别是原子的电离能和电子亲和能。这才是电负性的第一个绝对量度。
二是1978年Parr根据W. Kohn的密度泛函理论(DFT,属于量子统计力学)把电负性定义为
_{v}) (5) (5)
称为绝对电负性 [21]。这里μ是体系的化学势。这两种定义所以重要是因为物理意义明确,尤其是绝对电负性。在电子密度泛函理论之前,人们往往下意识地以为只有宏观体系才有化学势,没想到微观体系一样有化学势这个概念,两者之间没有鸿沟,是一个概念。式(5)给μ加个负号只是为了与Mulliken电负性的正负号大体一致而已。Mulliken电负性可以看成是Parr绝对电负性的近似(图1)。图1是Parr的绝对电负性χ、Mulliken电负性、化学势μ和Parr的绝对硬度η四个概念之间的几何联系,也许Parr就是由此从Mulliken电负性中悟出绝对电负性的真正含义的。
![单靠实验数据统计拟合能逼近真理吗? ——论“凭什么相信计算”之三-1]()
图1 Parr的绝对电负性χ、Mulliken电负性 、化学势μ,和Parr的绝对硬度η四个概念之间的几何联系 、化学势μ,和Parr的绝对硬度η四个概念之间的几何联系
Parr的绝对电负性χ所以能够一锤定音,是因为以下鲜明特点,赋予了它严格的物理意义[21]:
1)Parr的绝对电负性χ是一个融入在热力学、量子力学、和统计力学这个统一的理论中的概念。若要推翻它,就不得不推翻整个热力学、量子力学、和统计力学这个理论体系,而后者已经经历过整个20世纪物理科学考验过的。显然推翻这样的概念要远远比推翻任意一种唯象理论要难得多,是几乎不可能的。
2)Parr的绝对电负性概念已经不限于分子中的原子,只要是多电子体系,即有多个原子核和多个电子组成的微观体系。当然包括固体、液体。
3)1951年Sanderson提出过“电负性均衡原理”[23],当时仅仅是对Pauling电负性的一种猜想,无法严格证明。现在,根据热力学原理,化学势μ相等是多相体系平衡的严格条件。那么只要把Pauling电负性换成Parr的绝对电负性,即换成了化学势,Sanderson电负性均衡原理就立刻有了严格的证明。
至此,历时半个世纪关于电负性的疑惑尘埃落定。回过头看,大多数“改进”的套路都是加入与此定性有关的物理化学变量当自变量,再加入待定参数,然后用统计数学建立数学模型。只有Mulliken的电负性没有按照这个套路,而它却最接近最终正确的答案Parr电负性。Allen电负性的提出比Parr绝对电负性晚11年,可见Allen当时还没有觉察问题早已解决,他还没看出老套路是没有出路的。
总之,电负性概念的历史演化再次说明单凭归纳法,无论增加多少数据、拟合得多好,也是不可能逼近真理的。最后命中真理的临门一脚归功于理论物理学,即演绎法[21]。
§4 硬软酸碱原理的演化
第三个实例是硬软酸碱原理(HSAB原理)。那是1963年R.G. Pearson首先提出的[24,25]。迄今任何一本介绍化学原理的书中都可以找到这个定性的化学原理。就如气体的状态方程、电负性、Hammett方程研究中发生的历史演化一样,HSAB原理一问世,很多人马上想到要在数值上“定量化”,而不是Pearson原来的强、中、弱三档;企图通过引入参数和基本的物理化学变量来使HSAB原理表述为定量的数学表式[26]。
Pearson是位很有学术洞察力的学者。他看出走老路,单靠数据累积、归纳拟合把HSAB原理“定量化”是无益的数学游戏。所以他当时就回答说:HSAB原理不需要定量化。([25], p.242)当别人热衷于走老路企图把HSAB原理定量化时,Pearson却悄悄离开了他开辟的这块领地。
时间到了1982年,R G Parr在发展密度泛函理论的同时,继续探讨与绝对电负性有关的量子统计理论[21]:将体系能量E对体系电子总数N在基态处作Taylor展开
=E\left ( N \right )+\left ( \frac{\partial E}{\partial N} \right )_{v\left ( \vec{r} \right )}dN+\frac{1}{2!}\left ( \frac{\partial ^{2}E}{\partial N^{2}} \right )_{v\left ( \vec{r} \right )}\left ( dN \right )^{2}+...=E\left ( N \right )-\mu dN+\eta \left ( dN \right )^{2}+...)
,
其中一阶项系数就是-μ即χ;而Parr把二阶项系数定义为绝对硬度η(简称硬度),即
_{v\left ( \vec{r} \right )}=\frac{1}{2}\left ( \frac{\partial \mu }{\partial N} \right )_{v\left ( \vec{r} \right )}) 。 (6) 。 (6)
Parr看出化学势随体系电子数的变化实际上体现了电子云形变的容易程度,即软硬程度。
Parr特地将他得到的这个新概念带到加州,给Pearson看。这下Pearson看到了希望。式(6)无论从表述形式、物理意义或理论深度来看都远比他往日的理解深邃。他看出Parr这条路才是改进HSAB原理的正路。以前他苦于找不到出路,才无奈离开这个领域。现在“峰回路转疑无路,柳暗花明又一村”,Pearson欣然接受,与Parr两人一起发表了关于绝对硬度的开创性文章[27]。Pearson后来感慨地说:“我要感谢R G Parr,他使我重新燃起对HSAB原理的兴致” [28]。
接受了Parr的绝对硬度η概念之后,Pearson犹如希腊神话中的巨人Antaeus,一旦他的脚接触到母亲大地女神Gaea,又重新迸发出了巨大的力量。Pearson用新概念重新总结经验事实,1987年他猜测可能存在“最大硬度原理”,他说:“看来自然界有这样的规则,即分子的形成会使得分子显得尽可能地变硬。”[28] Pearson把这个猜想告诉了Parr。1991年Parr根据统计力学中的涨落理论和Onsager的涨落-回归假设给Pearson的最大硬度猜想提供了严格的证明[29]。从此,化学又增加了一个严格的原理;Pearson-Parr最大硬度原理。关于Onsager的涨落-回归假设和最大硬度原理的证明细节可参见拙著[11]的§10.2节和§16.1.5节。Onsager的涨落-回归假设是L Onsager(1903-1976)获得1968年诺贝尔化学奖的重要组成部分,可由此得到Onsager倒易关系,是不可逆热力学的基础之一。
回顾从绝对电负性到绝对硬度,两者之间通过体系能量对电子数之间的关系联系起来。绝对硬度又通过Onsager倒易关系联系到最大硬度原理。还可以再通过Onsager倒易关系与很多动力学系数,即与热扩散中的Soret效应、温差电动势中的Seeback效应、电致冷中的Peltier效应、压电效应等性质联系起来。物质性质之间如此四通八达的内在物理联系,有力地说明以上绝对电负性和绝对硬度的概念才真正体现了科学的客观真理性。如此由演绎法提升发现的概念有了质的变化,绝不是任何唯象理论比得上的。显然,即使将原来的唯象理论添加更多的实验数据、进一步拟合得多好,凭那样的老套路是走不出迷宫的,不可能逼近科学真理。
§5 唯象理论改进与统计模型
单凭实验数据拟合本质上是统计意义上的“改进”,它与在物理意义上的改进决不是一回事,不能相提并论。在这点上,统计数学的鼻祖、剑桥大学的Ronald A. Fisher教授(1890-1962)有一句极为精辟的话:“统计本质上是一种归纳推理。”[30]。换言之,那就是“Russell鸡”的单纯归纳推理[22]。Fisher的话已经说透了,即统计数学本身是严格的、无瑕可击的,但应用过程中有陷阱,有风险,使用者应该自己明白[31]。同一个统计数学方法可以拿来做科学计算,也可以拿来卜卦、算命。笔者在2004年的一次分子模拟学术会议上介绍过用偏最小二乘法做一个算命的多元线性统计模型,算出求卦者的月工资:设“算命”的线性统计模型为
 , (7) , (7)
其中y指月工资收入; 为50个参数,分别代表影响收入的可能因素:学历、时辰八字、职业、外语水平、血型、星座……等(姑且先不论其科学性,都量化为数字); 为50个参数,分别代表影响收入的可能因素:学历、时辰八字、职业、外语水平、血型、星座……等(姑且先不论其科学性,都量化为数字); 为对应的50个待定系数。先招募10位志愿者(样本),提供数据 为对应的50个待定系数。先招募10位志愿者(样本),提供数据) 10套。代入式(7),试求系数 10套。代入式(7),试求系数 。可是得到的只有10个线性方程,待求的系数 。可是得到的只有10个线性方程,待求的系数 却有50个。此类问题在线性代数中尚缺40个方程,无解。但却可用统计数学来解,如用偏最小二乘法(PLS)求解。其原理是通过自变量 却有50个。此类问题在线性代数中尚缺40个方程,无解。但却可用统计数学来解,如用偏最小二乘法(PLS)求解。其原理是通过自变量 与因变量y之间的相关分析找出影响最大的前10个自变量(主成分),其余自变量所对应的40个 与因变量y之间的相关分析找出影响最大的前10个自变量(主成分),其余自变量所对应的40个 均当作零。这样就可解出前10个 均当作零。这样就可解出前10个 系数。完成算命统计模型的建立。接着就可以摆摊算命。求卦者报出数据 系数。完成算命统计模型的建立。接着就可以摆摊算命。求卦者报出数据 ,代入式(7),就可电脑打印出月工资y值。 ,代入式(7),就可电脑打印出月工资y值。
笔者以此作反例,提醒科研要与算命划清界限。不能因为算命模型中有些自变量 如学历、外语水平与收入有明显的因果联系,即有“物理意义”,而与算命划不清界限。此类数学模型或黑箱方法只能在不得已时采用,不能乐此不疲。 如学历、外语水平与收入有明显的因果联系,即有“物理意义”,而与算命划不清界限。此类数学模型或黑箱方法只能在不得已时采用,不能乐此不疲。
非但算命,同理,只要安排无数种“实验测量 + 统计数学方法”的科研项目,岂不就可以解决不同领域的所有科研任务了吗?显然是荒谬的。
只要把涉及的统计数学原理一步不跳地演绎一遍,思考一番,就不难明白其中的道理。Fisher的话自然就明白了,明白统计意义的正确与物理意义的正确决不是一回事。我国学者首创的“灰箱理论”只能在世界工程界得到响应,而难以在科学界施展影响就是这个道理。工程界强调预言,科学界强调的却是解释或理解。两者存在质的区别。
物理学大师吴大猷教授特别注意到:“爱因斯坦十分强调不可能存在一种‘归纳的理论’,一切物理理论必定建立在演绎的基础上”[32]。可见这个观点在物理学界、数学界早已确立了,而要让今天的化学界接受它还颇费口舌。
在科学进步的道路中,引入参数往往用以暂时越过理论的困难。相当于物理模型和数学模型混合采用。值得注意:不同的参数还有层次不同的差别。Van der Waals方程中的参数a,b层次较浅;半经验量子化学计算的AM1、PM3方法中引入的参数(如原子电离势、电子亲和能等)的层次相比要深一些。电子密度泛函理论中近似处理交换-相关能泛函项 [ [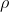 ]引入的参数其层次就相当深了(如最早的B3LYP、B3PW91泛函中的参数)。参数个数越少、层次越深的理论,其中物理模型相对于数学模型占的份量就愈重,其物理意义往往更清晰。 ]引入的参数其层次就相当深了(如最早的B3LYP、B3PW91泛函中的参数)。参数个数越少、层次越深的理论,其中物理模型相对于数学模型占的份量就愈重,其物理意义往往更清晰。
当然,必须承认以上讨论都是对无生命世界的,涉及有生命的问题不在此列。后者目前或许只能用QSAR之类的黑箱方法,那是因为目前在严密科学(exact science)的理论武库里(包括量子力学和统计力学等)还没有与Darwin生物进化对应的演化算符(evolution operator);已有的演化算符只是Newton或Liouville意义上的时间演化。
§6 von Neumann的大象•小结
2004年Freeman Dyson在《Nature》上发表了仅一页的回忆文章“与Erico Fermi的一次谈话”,副题为:一位极具直观洞察力的物理学家是如何把一个研究团队从歧路上挽救过来的[33]。文章说:1953年Dyson在理论中引入4个参数,计算出介子与质子的散射截面值与Fermi的实验值十分符合。于是,他兴冲冲搭灰狗长途车到芝加哥大学当面给Fermi过目。哪知Fermi不以为然,反倒讲了一个故事:“我记得我的朋友John von Neumann[34]曾经说过,用4个参数可以拟合出一头大象,再用第5个参数还可以让象鼻子摆起来。”谈话让Dyson很是沮丧。后来尽管文章发表了,但是Fermi的话深深地震撼了Dyson。甚至五十年后81岁高龄的他还觉得有必要把这段自己误入歧途并受到Fermi及时挽救的亲历故事讲给后来人听。这充分说明:任意引入参数以期提高结果,其统计意义上的准确性和物理意义的正确是根本不同的两回事。
2010年有三个较真的德国人要检验Fermi的故事。果然,他们用四个复参数用计算机成功模拟出大象的形态,又再引入一个复参数让象鼻子都动了起来[35]。这就更是验证了单靠追求数值上的准确,无助于了解其物理意义。那样的计算用于探索科学原理是不可信的,只能用于制作《侏罗纪》动画片、单纯的工程仿真等。
唯象理论是在不明事物内在原因的情况下,用统计数学方法从大量实验数据拟合得到的物理化学规律。在实验数据范围内,唯象理论对自然现象有描述或“预言”的能力,但没有解释、理解的能力。
科学的真正目的到底是预言世界还是解释世界呢?D. Deutsch教授的一番话值得深思:“理解并不取决于知道许多事实,而是依赖于正确的概念、正确的解释和正确的理论。一个相对简单而有一般性的理论可以覆盖无穷多的难以理解的事实。……预言事物或描述事物,不论多么准确,也和理解不是一回事。” Deutsch批评对待理论的一种常见态度,说:“……有些哲学家,甚至有些科学家,蔑视科学的解释作用。对他们来说,科学理论的基本目的,不是解释任何事情,而是预言实验结果,即科学理论的全部内容是它的预言公式。他们认为理论只要与预言结果一致,任何解释都是一样的,没什么好坏之分,甚至有没有解释无所谓,只要预言是正确的。”[36]。
本文从气体的状态方程、电负性和硬软酸碱理论三个科学实例的历史进程分析,试图给目前还在“改进”诸如Hammett方程的研究者参考。如果把这个历史进程比喻成迷宫,那么从已经走出迷宫的实例来看,站在迷宫的出口回头遥望迷宫的进口,可以让我们看到前进的方向。Dyson的历史教训极为深刻:单靠增加实验数据,归纳拟合,不断“改进”是做不到的;要尽量采用物理模型,不得已才暂时使用数学模型[37]。
参考文献
[1] 陈敏伯,“关于能量最低原理的误读:论‘凭什么相信计算’之二”,化学通报,2015, 78: 868.
[2] 陈敏伯,“热力学的公理体系:论‘凭什么相信计算’之一”,化学通报, 2013, 76: 388.
[3] L P Hammett. J. Am. Chem. Soc. 1937, 59: 96.
[4] 曹晨忠. 《有机化学中的取代基效应》,科学出版社,2003年,第69页;
H H Jaffe. Chem. Rev. 1953, 53: 191-261.
[5] M J S Dewar. The Molecular Orbital Theory of Organic Chemistry, New York: McGraw-Hill, 1969; 中译本:《有机化学分子轨道理论》,戴树珊等译,江元生校,科学出版社,1979年;第9.15节,第525页。
[6] W J Lyman, et al. Handbook of Chemical Property Estimation Methods: Environmental Behavior of Organic Compounds, Am. Chem. Soc, 1982; 中译本:《化学性质估算方法手册:有机化合物的环境性质》,许志宏等译,化学工业出版社,1991年。
[7] L Pauling. J. Am. Chem. Soc. 1932, 54: 3570;
L Pauling. The Nature of Chemical Bond, 3rd ed. Cornell University Press, Ithaca, 1960.
[8] P Atkins, J de Paula. Physical Chemistry, 9th ed. W.H. Freeman, 2010.
[9] http://en.wikipedia.org/wiki/Equation_of_state ;
沈惠川,《统计力学》,中国科大出版社,2011年;
[日]妹尾学,《化学公式手册》,科学出版社,1987年.
[10] J E Mayer, M G Mayer. Statistical Mechanics, New York: John Wiley, 1940;中译本:《统计力学》,高等教育出版社,1957年。
[11] 陈敏伯,《统计力学:理论化学用书》,科学出版社,2012年,§5.5节,§10.2节,§16.1.5节。
[12] 宋子良等,《范德瓦尔斯及其学派》,华中科技大学出版社,2002年,第218页。
[13] http://en.wikipedia.org/wiki/Electronegativity .
[14] R S Mulliken. J. Chem. Phys. 1934, 2: 782.
[15] W Gordy, Phys. Rev., 1946, 69: 604.
[16] 李世瑨,化学学报, 1957, 23: 234.
[17] A L Allred, E G Rochow. J. Inorg. Nucl. Chem. 1958, 5: 264.
[18] 高孝恢,化学学报, 1961, 27: 190.
[19] 温元凯,中国科技大学学报, 1974, 4: 145;
温元凯,邵俊,《离子极化导论》,合肥:安徽教育出版社,1985年.
[20] L C Allen, J. Am. Chem. Soc., 1989, 111: 9003.
[21] R G Parr, R A Donnelly, M Levy, W E Palke. J. Chem. Phys. 1978, 68, 3801;
R G Parr, W Yang. Density-Functional Theory of Atoms and Molecules, Oxford Science Publications, New York, 1989.
[22] “Russell鸡”是指英国数学家、数理逻辑学奠基人B. Russell(1872-1970)所著《哲学原理》第6章“归纳法”故事中的那只鸡。故事讲农夫养了一只鸡,每天清晨按时喂食,日久,鸡从中总结出规律:凡清晨那熟悉的脚步声走来,总能得从那农夫手中得到食物。可是有一天清晨农夫走来,非但不给食,却一把抓起鸡脖子要宰。那只“Russell鸡”临死前才明白牠总结规律的方法是不可靠的,可为时已晚。曾经掀起第三次数学危机的Russell无非是让人们明白:那只鸡认识规律的方法就是我们人类认知采用的归纳法(induction);提醒人们不能把实验科学建立在单靠归纳法的基础上。认识世界所依靠的两大方法,归纳法和演绎法(deduction),缺一不可。看来最早认识到单凭实验数据拟合不能逼近真理的人是B. Russell。他又是位思想家,他的《西方哲学史》荣获1950年的诺贝尔文学奖。
[23] R T Sanderson, Science, 1951, 114: 670-672.
[24] R G Pearson. J. Am. Chem. Soc. 1963, 85: 3533.
[25] R G Pearson. Hard and Soft Acids and Bases. Dowden, Hutchson, & Ross, Inc., 1973.
[26] 何子乐.《有机化学中的硬软酸碱理论》,科学出版社,1987年.
[27] R G Parr, R G Pearson. J. Am. Chem. Soc. 1983, 105: 7512.
[28] R G Pearson. Recent Advances in the Concept of Hard and Soft Acids and Bases, J. Chem. Edu., 1987, 64: 561.
[29] R G Parr, P K Chattaraj. J. Am. Chem. Soc. 1991, 113: 1854.
[30] R A Fisher. Statistical Methods and Scientific Inference, Oliver & Boyd, 1956;
张尧庭,“概率概念的发展和争论”,《数学与文化》,邓东皋等编,北京大学出版社,1990年;第359页。
[31] D Huff. How to Lie with Statistics, New York: W. W. Norton & Co., 1982; 第一个中译本:《怎能用统计撒谎》,中国统计出版社,1986年;第二个中译本:《统计陷阱》,上海财经大学出版社,2003年。
[32] 吴大猷,《物理学的历史和哲学》,金吾伦, 胡新和译. 中国大百科全书出版社 , 1997。
[33] Dyson, F., Nature 2004, 427: 297.
Freeman Dyson(1923-)是位少年得志的美籍英国数学家和理论物理学家。1949年还在师从Hans Bethe读研究生期间就证明了Schwinger与朝永振一郎的变分法和Feynman的路径积分法两者是等价的,为量子电动力学的确立作出了重要贡献,蜚声四海。后三位因量子电动力学的建立荣获1965年的诺贝尔物理学奖。传说,因人数限制Dyson才与诺奖失之交臂。
[34] John von Neumann(1903-1957)美籍匈牙利数学家。1927年出版名著《量子力学的数学基础》解决了Hilbert第6个问题“物理学公理化”中的一部分。他又是博弈论、计算机科学的奠基人。
[35] Mayer, J., et al., “Drawing an elephant with four complex parameters”, Am. J. Phys. 2010, 78: 648;
邢志忠,科学世界,2014年, 6月号, 110。
[36] D Deutsch. The Fabrics of Reality, Allen Lane, 1997; 中译本:《真实世界的脉络》,广西师范大学出版社,2002.
David Deutsch(1953- ),1998年Dirac奖得主,量子计算的奠基人,牛津大学物理学教授。
[37] J C Taylor. Hidden Unity in Nature’s Laws, Cambridge University Press, 2001; 中译本:《自然规律中蕴蓄的统一性》,北京理工大学出版社,2004,第8页.
【注:附上PDF版文件】![单靠实验数据统计拟合能逼近真理吗? ——论“凭什么相信计算”之三-2]()
往期无机物化专家(mbchen)交流帖 能量最低原理到底指什么意思
http://muchong.com/bbs/viewthread.php?tid=9937320&fpage=1
关于能量最低原理的误读
http://muchong.com/bbs/viewthread.php?tid=10536152&fpage=1
热力学的公理体系 ——论“凭什么相信计算”之一
http://muchong.com/bbs/viewthread.php?tid=10537196&fpage=1
关于能量最低原理的误读 ——论“凭什么相信计算”之二
http://muchong.com/bbs/viewthread.php?tid=10536152&fpage=1
[ Last edited by 冰点降温 on 2016-8-5 at 09:59 ] |
» 本帖附件资源列表
» 收录本帖的淘帖专辑推荐
» 本帖已获得的红花(最新10朵)
» 猜你喜欢
|
 【官方】无机物化
【官方】无机物化 个人收藏
个人收藏 暖通
暖通 [武汉纺织大学]材料学院[姜伟团队名称]课题组-2026年硕士研究生调剂信息
已经有0人回复
[武汉纺织大学]材料学院[姜伟团队名称]课题组-2026年硕士研究生调剂信息
已经有0人回复
 武汉纺织大学-材料科学与工程学院-姜伟杰青团队-招调剂
已经有0人回复
武汉纺织大学-材料科学与工程学院-姜伟杰青团队-招调剂
已经有0人回复
 有机高分子材料论文润色/翻译怎么收费?
已经有204人回复
有机高分子材料论文润色/翻译怎么收费?
已经有204人回复
 生物医学材料前沿科普:低密度、高孔隙率的“多孔微球”核心技术与应用全景
已经有1人回复
生物医学材料前沿科普:低密度、高孔隙率的“多孔微球”核心技术与应用全景
已经有1人回复
 湖南工业大学周贵寅组招生物医学工程(学硕07、08都行)、生物与医药(专硕08)硕士生
已经有0人回复
湖南工业大学周贵寅组招生物医学工程(学硕07、08都行)、生物与医药(专硕08)硕士生
已经有0人回复
 湖南工业大学周贵寅组招生物医学工程(学硕07、08都行)和生物与医药(专硕08)硕士生
已经有0人回复
湖南工业大学周贵寅组招生物医学工程(学硕07、08都行)和生物与医药(专硕08)硕士生
已经有0人回复
 考研求调剂
已经有0人回复
考研求调剂
已经有0人回复
 2026年中南民族大学化学与材料科学学院调剂公告
已经有40人回复
2026年中南民族大学化学与材料科学学院调剂公告
已经有40人回复
 武汉纺织大学招生物材料、化学、高分子相关专业硕士生
已经有0人回复
武汉纺织大学招生物材料、化学、高分子相关专业硕士生
已经有0人回复
 武汉纺织大学招生物材料、化学、高分子相关专业硕士生-另有少量士兵计划可咨询
已经有0人回复
武汉纺织大学招生物材料、化学、高分子相关专业硕士生-另有少量士兵计划可咨询
已经有0人回复
 [武汉纺织大学]材料学院[姜伟团队名称]课题组-2026年硕士研究生调剂信息
已经有0人回复
[武汉纺织大学]材料学院[姜伟团队名称]课题组-2026年硕士研究生调剂信息
已经有0人回复
















 回复此楼
回复此楼 流火
流火



 5
5